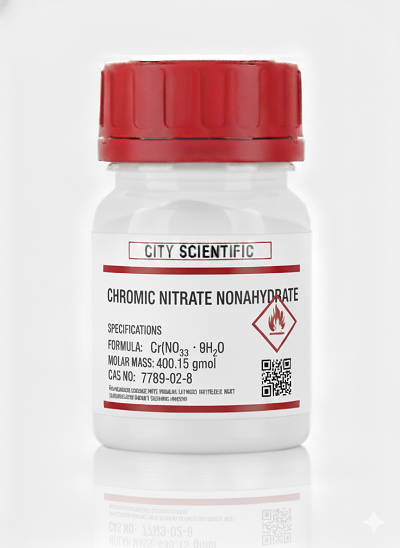
- Catalyst preparation: It is widely used to prepare chromium-based catalysts for applications in petrochemicals and organic synthesis.
- Textile and leather industries: The compound serves as a mordant in textile printing and dyeing processes, helping to fix dyes to fabrics. It is also used in leather tanning.
- Electroplating: It can be used as a source of chromium ions in electroplating processes to enhance the corrosion resistance and appearance of metal surfaces.
- Coordination complexes: The compound is a common starting material in academic laboratories for the synthesis of chromium coordination complexes.
- Corrosion inhibitor: It is used as a corrosion inhibitor in certain applications.
- Analytical chemistry: It is employed as a reagent in various analytical methods.
Safety and handling precautions
- Hazard identification: The substance is classified as a hazardous chemical, with a signal word of “Warning”.
- Oxidizer: It may intensify a fire and presents an explosion risk when in contact with combustible materials.
- Health risks: It can cause serious eye irritation and may be harmful if swallowed or inhaled. Skin contact may cause an allergic reaction.
- Personal protective equipment: Wear protective gloves, goggles, and clothing during handling. Use a respirator if dust is formed.
- Storage: Store in a cool, dry, well-ventilated area, away from heat, sparks, and incompatible materials.
- Environmental hazards: It is toxic to aquatic organisms and should not be released into the environment.
- Disposal: Waste should be classified and disposed of according to local and national hazardous waste regulations.